Part Of: Neural Oscillators sequence
Related To: Habit as Action Chunking
Content Summary: 1800 words, 9 min read
Overview of Spatial Navigation
The hippocampus is centrally important for spatial navigation. Neuroscientists have discovered specialized cell types in the medial entorhinal cortex (MEC), including border cells, head-direction cells, and grid cells.
Grids are actually organized hierarchically, with grids growing in size along the dorsal-ventral axis within MEC. But grid size does not increase continuously, rather there are precisely four interlocked grids (Stensola et al 2012). The ratio between grid distances is consistently 1.42, which may be optimally efficient (Mathis et al 2012).
MEC navigational cells allow for the complex behavior of path integration. An ant leaving its nest will search for food by exploring the environment; once it decides to return, it integrates all of its various exploratory movements (each vector is reconstructed from direction, speed, and duration cells) into a single, precise return trajectory. If you abruptly move the ant a couple meters before it returns, it will follow the same return vector, and exhibit confused-search behavior when it cannot find the nest.
But the hippocampus also supports another, more objective approach: allocentric navigation. Essentially, landmarks are used to orient one’s environment.
Place cells also exhibit a reward gradient, perhaps stemming from interactions with the basal ganglia. This reward gradient is significant in reinforcement learning (RL) models.
These independent navigational systems rely on two different representations of the body: egocentric (me-centered) and allocentric (world-centered) perspectives. These representations can come apart in clinical out-of-body experiences. For example, clever uses of VR to disrupt egocentric processing leads to a reduction in activity in egocentric parts of the hippocampus (Bergouignan et al 2015)
Overview of Oscillation
Neural oscillations are a central organizational principle by which the brain coordinates different neural ensembles (Buzsaki 2010). In the following image (a), each row is a different neuron. With these multi-unit recordings, you’ll find that certain groups of neurons consistently fire together (gray ovals) in the trough of the theta cycle.
There are at least ten different oscillators with unique cognitive functions.
Relevant to today’s post, we have,
- Gamma oscillations (30-100 Hz, 10-30ms window) are ubiquitous across cortex and associated with perceptual binding (Engel & Singer 2001).
- Theta oscillations (4-10 Hz, 100-250ms window) are not as global, but have been detected in hippocampus, MEC, medial prefrontal cortex and ventral striatum (Drieu & Zugaro 2019).
Gamma oscillators can be phase locked with theta, with ~7 gamma cycles nested within theta. Theta and gamma exhibit phase-amplitude coupling (PAC), an example of the more general mechanism of cross-frequency coupling (CPC). This theta-gamma neural code is important for learning and memory (Lisman & Jensen 2013).
Theta Sequences and Phase Precession
In the brain, behavior is represented with at least two different timescales.
- During locomotion, place cells fire in behavioral sequences that change approximately every 1-2s (T).
- The same trajectories are simultaneously played out in theta sequences every 100-200ms (tau).
As the rat moves, place cells fire at progressively earlier phases within theta cycle. This phase precession was first noticed by O’Keefe & Recce (1993).
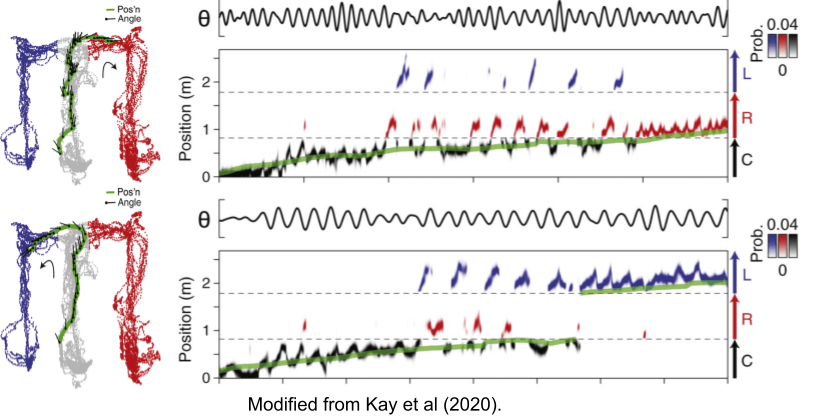
The ~7 gamma bursts within each theta rhythm seems to encode a planning trajectory, or sweep, of its upcoming behaviors. In Wikenheiser & Redish (2015), rats could either stop at a feeder (upper left location) or continue moving through the track. Their theta sequences predicted their behavioral choice, well ahead of time. A similar result was found by Gupta et al (2012).
What happens when the rat is still making up its mind? Theta sweeps examine both trajectories within a single trial (Johnson & Redish 2007; Kay et al 2020).
Jezek et al (2011) used flickering to induce confusion whether a rat was located in one of two environments. They found that theta sequences switched between either environment, but within-sequence representations were consistent. This suggests that theta sequences may represent cognitive primitives.
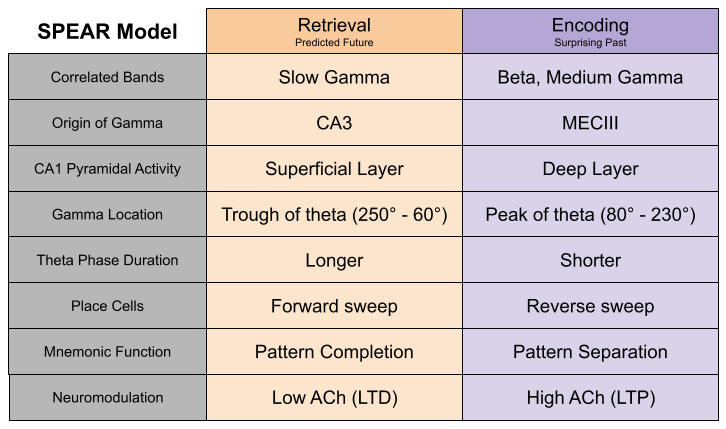
The SPEAR model
We have detected sub-processes within theta sequences. Hasselmo et al (2002) noticed several physiological differences that occur at the peak versus the trough of the theta cycle. They proposed that peak of the theta phase is involved with the encoding of information, while the trough features a retrieval of information. These phases correspond to the pattern separation versus pattern completion, respectively. Notably, long term potentiation (LTP) was much stronger at theta peak. This is largely driven by acetylcholine, which is expressed more strongly during encoding than retrieval (Hasselmo 2006).
Belluscio et al (2012) found that slow gamma (30-50 Hz) and medium gamma (50-90 Hz) exclusively occur in the trough and peak of theta, respectively. Schomberg et al (2014) replicated this result. Wang et al (2020) note that beta oscillations occur only during theta trough.
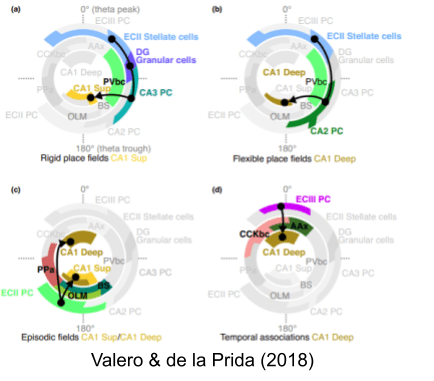
Navas-Olive et al (2020) associated activity in superficial and deep CA1 pyramidal neurons with trough and peak of theta, respectively. Their lab has posited four sub-circuits which fire at distinctive locations in theta phase.
Phase precession often reveals prospective choices. But neuroscientists have long noted the existence of retrospective sweeps, theta sequences of locations behind the animal. Bieri et al (2014) have linked slow and slow gamma to prospective and retrospective sweeps, respectively. As we saw in the image above, prospective sweeps simulate possible futures, while retrospective sweeps encode the past (Kay et al 2020).
Here are the properties of the Separate Phases of Encoding And Retrieval (SPEAR) model.
The SPEAR Model explains several behavioral results.
Takahashi et al (2014) found that during a fixation cue, where a rat is forced to rely on spatial memory to make a movement decision, there is an abrupt shift to slow gamma emerging from CA3. This is consistent with its hypothesized function of retrieval.
In novel environments, rats tend to move quickly and non-linearly. As an environment becomes familiar, speed slows down but becomes more linear. Kemere et al (2013) show that fast gamma is expressed at higher speeds, and vice versa. This may reflect the process by which as an animal familiarizes itself with an environment, its cognitive operations increasingly rely on memory retrieval. This result was replicated in Zheng et al (2016).
Relation to Working Memory
Long ago, Sternberg (1966) reported a linear relationship between recall latency and the number of memorized items, which suggested that the list was serially scanned at a rate of 20-30ms per memory item. Similarly, Miller (1955) also collated many different experiments showing a ceiling of recall of about 7 items (but the limit becomes 4 items when rehearsal is removed; Cowan 2000).
These constraints on working memory are suggestive: the 20-30ms scan time aligns with the gamma cycle, and there are about 7 gamma cycles within a theta sequence (Lisman & Idiart 1995). The ratio of theta to gamma may even correlate with WM span (Kaminski et al 2011), although this data is fairly uncertain.
Theta power increases systematically with working memory load (Jensen & Tesche 2002). Theta does not occur uniformly across cortex during WM tasks, but is localized to specific sites (Raghavachari et al 2001). Theta frequency decreases during periods of high load, consistent with more representations requiring longer theta sequences (Axmacher et al 2010; but see Moran et al 2010).
Heusser et al (2016) found that maximum gamma power for the memory items occured at distinct locations in theta phase, but only when the subject was able to remember the sequence correctly. Theta also appears conducive to binding together sequences across different modalities. This was replicated in Reddy et al (2021). Clouter et al (2017) found that when audio and video clips were synchronized to theta oscillations (but not other frequency bands), recall accuracy substantially improved.
Roux & Uhlhaas (2014) review studies that explore neural oscillations during WM maintenance. They found that theta activity occurs preferentially in tasks that involve the sequential items, whereas alpha oscillations tend to occur during tasks that require simultaneous maintenance of visuospatial information. They propose to identify theta-gamma binding with one of the subcomponents of working memory: the phonological loop.
Griffiths et al (2021) found that theta-gamma PAC (memory consolidation) arose after reductions in alpha/beta power (sequence perception), indicative of a two-stage retention process.
Until next time.
References
- Axmacher et al (2010). Cross-frequency coupling supports multi-item working memory in the human hippocampus
- Bieri et al (2014). Slow and fast gamma rhythms coordinate different spatial coding modes in hippocampal place cells
- Bullicino et al (2012). Cross-Frequency Phase–Phase Coupling between Theta and Gamma Oscillations in the Hippocampus
- Buzsaki (2010). Neural syntax: cell assemblies, synapsemblies, and readers
- Canolty et al (2006). High gamma power is phase-locked to theta oscillations in human neocortex
- Clouter et al (2017). Theta Phase Synchronization Is the Glue that Binds Human Associative Memory
- Colgin et al (2009). Frequency of gamma oscillations routes flow of information in the hippocampus
- Cowan (2000). The magical number 4 in short-term memory: A reconsideration of mental storage capacity
- de Almeida (2009). The Input-Output Transformation of the Hippocampal Granule Cells: From Grid Cells to Place Fields
- Drieu & Zugaro (2019). Hippocampal Sequences During Exploration: Mechanisms and Functions
- Engel & Singer (2001). Temporal binding and the neural correlates of sensory awareness.
- Griffiths et al (2021). Disentangling neocortical alpha/beta and hippocampal theta/gamma oscillations in human episodic memory formation
- Gupta et al (2012). Segmentation of spatial experience by hippocampal theta sequences
- Hasselmo et al (2002). A proposed function for hippocampal theta rhythm: separate phases of encoding and retrieval enhance reversal of prior learning
- Hasselmo (2006). The Role of Acetylcholine in Learning and Memory
- Hasselmo & Stern (2013). Theta rhythm and the encoding and retrieval of space and time.
- Heusser et al (2016). Episodic sequence memory is supported by a theta–gamma phase code
- Jensen & Tesche (2002). Frontal theta activity in humans increases with working memory load in a working memory task.
- Jezek et al (2011). Theta-paced flickering between place-cell maps in the hippocampus
- Johnson & Reddish (2007). Neural ensembles in CA3 transiently encode paths forward of the animal at a decision point
- Kaminski et al (2011) Short term memory capacity predicted by theta to gamma cycle length ratio.
- Kemere et al (2013). Rapid and continuous modulation of hippocampal network state during exploration of new places
- Lisman & Idiart (1995). Storage of 7+/-2 short-term memories in oscillatory subcycles
- Lisman & Jensen (2013). The theta-gamma neural code
- Mathis et al (2012). Optimal Population Codes for Space: Grid Cells Outperform Place Cells
- Miller (1955). The Magical Number Seven, Plus or Minus Two: Some Limits on Our Capacity for Processing Information
- Moran et al (2010). Peak frequency in the theta and alpha bands correlates with human working memory capacity.
- Navas-Olive et al (2020). Multimodal determinants of phase-locked dynamics across deep-superficial hippocampal sublayers during theta oscillations
- O’Keefe & Recce (1993). Phase relationship between hippocampal place units and the EEG theta rhythm
- Penttonen & Buzsaki (2003). Natural logarithmic relationship between brain oscillators
- Raghavachari et al (2001). Gating of human theta oscillations by a working memory task
- Reddy et al (2021). Theta-phase dependent neuronal coding during sequence learning in human single neurons
- Roux & Uhlhaas (2014). Working memory and neural oscillations: alpha–gamma versus theta–gamma codes for distinct WM information?
- Schonberg et al (2014). Theta phase segregation of input-specific gamma patterns in entorhinal-hippocampal networks
- Shrivalkar et al (2010). Bidirectional changes to hippocampal theta-gamma comodulation predict memory for recent spatial episodes
- Srinivasan (2015). Where paths meet and cross: navigation by path integration in the desert ant and the honeybee
- Stensola et al (2012). The entorhinal grid map is discretized
- Sternberg (1966). High-Speed Scanning in Human Memory
- Takahashi et al (2014). Theta phase shift in spike timing and modulation of gamma oscillation: a dynamic code for spatial alternation during fixation in rat hippocampal area CA1
- Valero & de la Prida (2018). The hippocampus in depth: a sublayer-specific perspective of entorhinal-hippocampal function.
- Wang et al (2020). Alternating sequences of future and past behavior encoded within hippocampal theta oscillations.
- Wikenheiser & Redish (2015). Hippocampal theta sequences reflect current goals
- Zheng et al (2015). The relationship between gamma frequency and running speed differs for slow and fast gamma rhythms in freely behaving rats
- Zheng et al (2016). Spatial Sequence Coding Differs during Slow and Fast Gamma Rhythms in the Hippocampus