Part Of: Biology sequence
Content Summary: 1800 words, 9 min read.
The Disposable Soma
Many people interpret aging as an inevitability: heat death is coming for us all. And it is true that most organisms experience senescence, i.e., age-related deterioration (Nussey et al 2012), even some bacteria (Ackermann et al 2003).
But some organisms experience negligible senescence (e.g., hydra Martinez 1998 or rockfish Finch 2009). Why isn’t negligible senescence the norm? As Williams (1957) put it,
It is remarkable that after a seemingly miraculous feat of morphogenesis, a complex metazoan should be unable to perform the much simpler task of merely maintaining what is already formed.
A clue: organisms rarely die from old age. Rather, environmental mortality (accidents, predation, starvation, disease, cold, etc) usually strike well before senescence does (Finch 1990). Why invest in maintaining a body that will be dead anyway for other reasons?
For example, 90% of wild field mice die in their first year (Phelan and Austad 1989), typically from cold. The three-year lifespan potential of the mouse is sufficient for its actual needs in the wild, and yet it is not excessive.
Consider the trade-offs associated with energy allocation. Because energy is scarce, the mouse will benefit by investing any spare energy into thermogenesis or reproduction, rather than better somatic maintenance, even though this means that damage will eventually accumulate to cause aging.
In metazoans, we saw cellular differentiation between somatic cells (which could specialize to ultimately become muscle, bone, etc) and germ cells (which promote reproduction). The germ line requires biological immortality: individual germ cells can die, but the lineage cannot be allowed to deteriorate. Germ cells accomplish this in part by better maintenance: for example, the protective enzyme telomerase only exists in germ cells and in certain adult stem cells. Once embryonic stem cells differentiate into somatic cells, there is a generalized downregulation of cell maintenance systems (Saretzki et al 2004).
This is the disposable soma theory of aging (Kirkwood 1977).
Aging vs Mortality
The idea that intrinsic longevity is tuned to the prevailing level of extrinsic mortality is supported by extensive observations on natural populations (Ricklefs 1998).
It’s not just observational evidence.
- Stearns et al (2000) imposed a high mortality regime on one group of fruit flies, and observed that their experimental group lived a shorter life and a reduced age of sexual maturity.
- In a natural experiment, opossums naturally evolving on a predator-free island lived longer than their phylogenetic siblings on a normal-predation population (Austad 1993).
Differences in mortality explains a large fraction of lifespan variance between species. For example, flying species enjoy weaker senescence than non-flying species, due to the shelter against mortality (Healy et al 2014). The lifespan-extending effect of mortality reduction is also seen…
- … in arboreal species (Shattuck & Williams 2010),
- … in fossorial species (Healy et al 2014),
- … in species that hibernate (Turbill et al 2011)
- … in species that evolve protective shells (Phillip & Abele 2010)
In most species, males senesce faster than females (Brooks & Garrett 2017). This is considered to be a direct investment of the male involvement in intrasexual (and intersexual) competition (Bonduriansky et al 2008), and the resultant increase in male mortality.
We might distinguish between two kinds of environmental mortality:
- Non-selective mortality (e.g., the onset of winter) which strikes at random
- Selective mortality (e.g., predation) which preferentially strikes certain demographics
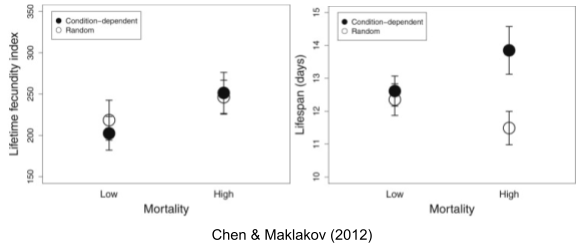
The above experimental results above relate to non-selective mortality. But Abrams (1993) has shown that evolutionary predictions can be considerably altered when density dependence is taken into account; selective mortality affects per capita resource availability. Chen & Maklakov (2012) found that lifespan decreases with random mortality, but increases with condition-dependent mortality (application of heat shock: only especially robust worms survive).
Aging vs Growth
Growth also drives senescence, because growth affects fecundity. Unceasing, indeterminate growth allows increasing fecundity with age. Indeed, senescence is negligible in organisms displaying indeterminate growth (Vaupel et al. 2004). But senescence is the rule in determinate growth organisms such as birds and mammals (Nussey et al. 2013).
Experimentally induction of catch-up growth reduces lifespan (Lee et al 2013).
In fact, growth rate is composed of two biologically distinct mechanisms. Development time is differentiation of the soma; under sexual selection during scramble competition (Andersson 1994). Growth is an increase in mass. When these are decoupled, only development time was found to be coupled to longevity (Lind et al 2017).
Many studies suggest that the hypothalamus-pituitary-somatotropic (HPS) axis mediates the tradeoff between growth and senescence (Dantzer & Swanson 2012). Fast species tend to have higher plasma insulin-like growth factor (IGF-1) than slow species (Swanson and Dantzer 2014; but see Stuart and Page 2010).
Two Genetic Theories of Aging
90% of wild rats die from cold in their first year, but in a protected laboratory environment those same rats live for three years, before dying of age-related diseases. From an evolutionary biologist’s perspective, senescence can evolve because selection gradients on mortality and fertility decline with age. Modeling by Hamilton (1966), and extended in Charlesworth (1994), found that selection is simply much weaker in old age; it casts a selection shadow.
Medawar (1952) noted the similarities between aging and Huntington’s disease. If the disease is inherited and always fatal why hasn’t natural selection expunged the responsible alleles from our gene pool? The answer, of course, has to do with the fact that Huntington’s disease typically strikes late in life history. He wrote “the force of natural selection weakens with increasing age… if a genetic disorder happens late enough in life, its fitness consequences may be completely unimportant”.
This is the mutation accumulation theory of senescence (MATS). It appeals to the ubiquity of pleiotropy: one gene can produce multiple, different effects across the life course.
Williams (1957) proposed the antagonistic pleiotropy theory of senescence (APTS). Rather than neutrality, some age-related genes are selected for their benefits to early life, despite those very same genes being deleterious later.
There are important differences between these two theories. Because mutations occur randomly, MATS predicts that aging will be fairly idiosyncratic to that lineage; whereas APTS suggests that the mechanisms underlying aging are comparatively more shared. If APTS is true, biogerontology research on model organisms should prove more informative to human therapeutics.
IGF-1 genes promote growth yet inhibit somatic maintenance. They are excellent candidates for antagonistic pleiotropy, and showcase how the disposable soma theory can bridge genetic and microbiological perspectives.
Some weak evidence has been found for MATS. In contrast, the evidence for APTS is fairly strong (Austad & Hoffman 2018), including identification of dozens of candidate genes. When these genes are knocked out, such as an IGF receptor gene in C elegans, the resultant stains live longer, but ultimately disappear due to a small reduction in early life fertility (Jenkins et al 2004). William’s APTS model generated nine predictions about senescence; today, six of these have found strong support (Gaillard & Lemaitre 2017).
The Search For Mechanism
A quote from Kirkwood (2005)
One oddity about aging is its inherent complexity. Almost every aspect of an organism’s phenotype undergoes modification with aging, and this phenomenological complexity has led, over the years, to a bewildering proliferation of ideas about specific cellular and molecular causes.
In the disposable soma theory, senescence is caused by failures to maintain the soma. But which specific mechanisms are involved? Microbiologists have not yet achieved consensus of causal mechanism. But they have achieved a consensus list of candidates: Lopez-Otin et al (2013) motivates nine hallmarks of aging.
Perhaps someday we will have therapeutics that effectively promote somatic maintenance.
Until next time.
References
- Abrams (1993). Does increased mortality favor the evolution of more rapid senescence?
- Ackermann et al (2003). Senescence in a bacterium with asymmetric division
- Andersson (1994). Sexual selection.
- Austad (1993). Retarded senescence in an insular population of Virginia opossums
- Austad & Hoffman (2018). Is antagonistic pleiotropy ubiquitous in aging biology?
- Bonduriansky et al (2008). Sexual selection, sexual conflict and the evolution of ageing and life span
- Brooks & Garrett (2017). Life history evolution, reproduction, and the origins of sex-dependent aging and longevity
- Charlesworth (1994). Evolution in Age-Structured Populations
- Chen & Maklakov (2012). Longer Life Span Evolves under High Rates of Condition-Dependent Mortality
- Cole (1954). The population consequences of life history phenomena
- Danson & Swanson (2012). Mediation of vertebrate life histories via insulin-like growth factor-1.
- Finch (1990). Longevity, senescence and the genome
- Finch (2009). Update on slow aging and negligible senescence – a mini-review
- Gaillard & Lemaitre (2017). The Williams’ legacy: A critical reappraisal of his nine predictions about the evolution of senescence
- Hamilton (1966) The moulding of senescence by natural selection
- Healy et al (2014). Ecology and mode-of-life explain lifespan variation in birds and mammals
- Jenkins et al (2004). Fitness cost of extended lifespan in Caenorhabditis elegans
- Jones et al (2008). Senescence rates are determined by ranking on the fast-slow life-history continuum.
- Kirkwood (1977). Evolution of ageing
- Kirkwood (2005). Understanding the odd science of aging
- Lee et al (2013). Experimental demonstration of the growth rate–lifespan trade-off
- Lind et al (2017). Slow development as an evolutionary cost of long life
- Lopez-Otin et al (2013). Hallmarks of aging
- Martinez (1998). Mortality patterns suggest lack of senescence in hydra.
- Medawar (1952). An unsolved problem of biology.
- Nussey et al (2013). Senescence in natural populations of animals: widespread evidence and its implications for bio-gerontology
- Omholt & Amdam (2004). Epigenetic regulation of aging in honeybee workers
- Phelan and Austad (1989). Natural selection, dietary restriction and extended longevity
- Phillip & Abele (2010). Masters of Longevity: Lessons from Long-Lived Bivalves – A Mini-Review
- Ricklefs (2010). Embryo development and ageing in birds and mammals
- Saretzski et al (2004). Stress Defense in Murine Embryonic Stem Cells Is Superior to That of Various Differentiated Murine Cells
- Shattuck & Williams (2010). Arboreality has allowed for the evolution of increased longevity in mammals.
- Stearns et al (2000) Experimental evolution of aging, growth, and reproduction in fruitflies.
- Stenvinkel & Shiels (2019). Long-lived animals with negligible senescence: clues for ageing research
- Stuart & Page 2010). Plasma IGF-1 is negatively correlated with body mass in a comparison of 36 mammalian species
- Swanson & Dantzer (2014). Insulin-like growth factor-1 is associated with life-history variation across Mammalia
- Turbill et al (2011). Hibernation is associated with increased survival and the evolution of slow life histories among mammals.
- Vaupel et al (2004) The case for negative senescence
- Wensink et al (2017). The rarity of survival to old age does not drive the evolution of senescence
- Williams (1957). Pleiotropy, natural selection and evolution of senescence